After that Deepmind post, lots of people reached out for an update on what *was* happening in the world of NKH Research in London, and I thought I’d share. It’s important to know that the money we’re raising is having a huge effect on the research done here in the UK. While I can’t speak for the other researchers (Dr Van Hove or Dr Katsuri Halder specifically), I do feel it’s important to note THERE IS RESEARCH happening (despite what that recent article by the Clemson World Magazine would have you believe).
There is A LOT of research happening, and it’s my genuine belief that Prof. Nick is the closest to meaningful clinical trial, with a treatment that will effect the most kids. But I’m getting ahead of myself.
Essentially, the team under Prof. Nick Greene is trying to do two things:
1 – understand more about how NKH works, and the effects it has
2 – discover potential treatments for NKH
Goal 1: Understand more about how NKH works, and the effects it has
There’s so much we don’t know – the carbon folate system (within which the glycine cleavage system lives) is really really complex. There are hundreds of amino acids – glycine included – which are split and joined and run through so many different processes. Super complex. Have you seen what that looks like? Here it is:
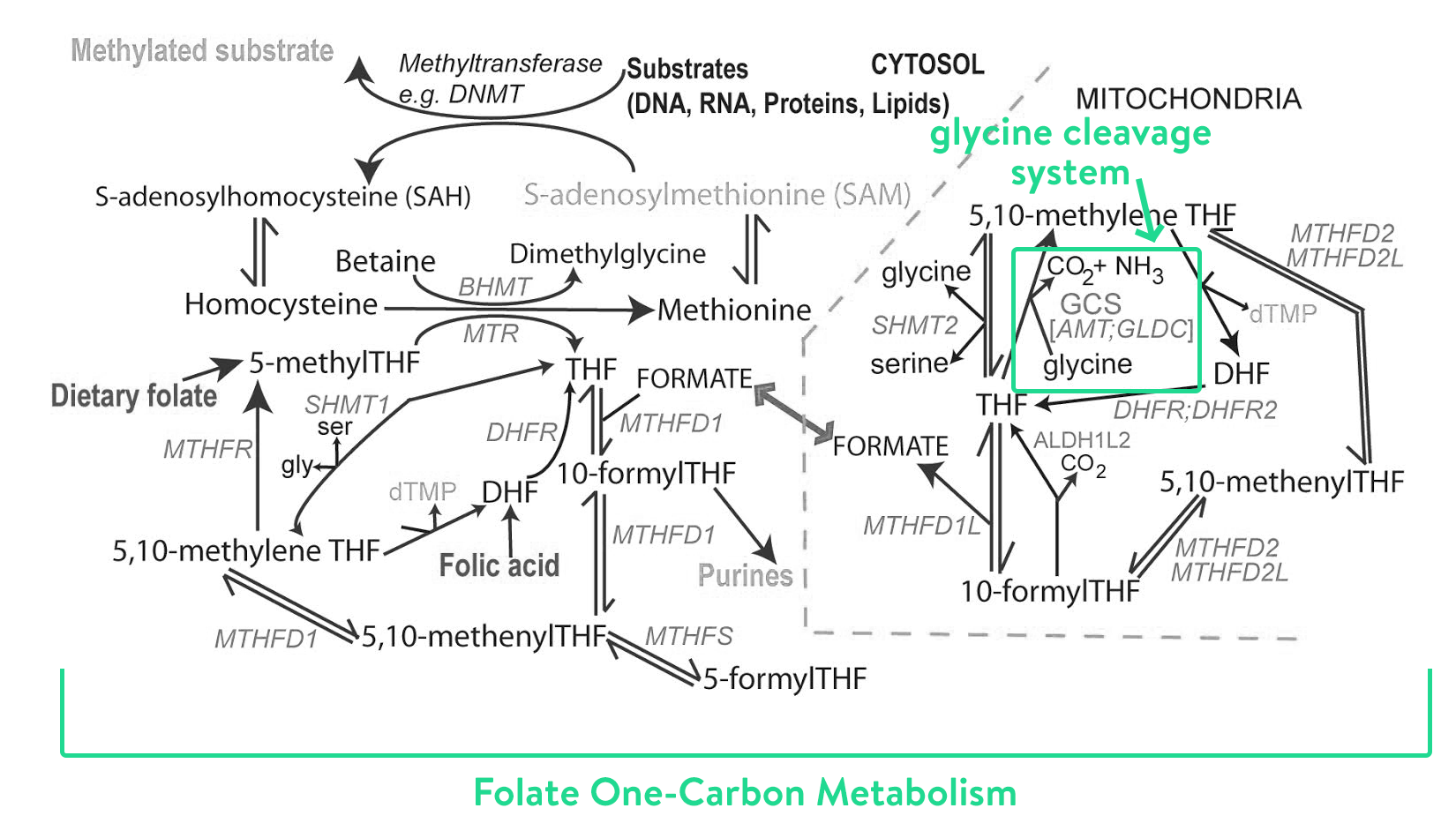
Source: Prof. Nick Greene, UCL
Truth: we don’t fully understand the how the whole system works, or all the roles glycine plays. This means it’s even harder to understand the knock on effects in a child with NKH. In the last year or so, Prof. Nick and his team have been using mass spectrometry (a really fancy science-y way to measure amino acids as they are processed) to follow glycine, and around 900 other different molecules in the brain and liver at different stages of NKH. They’ve been doing this in two different models – mice models, and in little cell samples grown from patients with NKH.
They’ve followed literally thousands of metabolic pathways. I had no idea that glycine went on to do so many things. It even joins up with other amino acids, like hexanoylglycine or propionylglycine or N-octagoylglycine (there are so, so many more). With excess levels of glycine, you can bet that all these other amino acids also have excess levels, which causes a knock on effect.
This small project alone – identifying all the metabolic pathways of glycine – costs £50-100k per year, but here’s the thing – they know more than they ever have before about NKH, giving insight into what’s happening with our kids. This is huge! It also speeds up the process, because it gives the research team clues about where in the process a treatment might work best. Knowing that means a faster route to clinical trials.
Which is everything – obviously we all want a safe and effective treatment for NKH. Which brings us to the treatment section.
Goal 2: Discover potential treatments for NKH
To get the treatment from research to people with NKH is a bit of a process.
First, they need to show ‘proof of concept’ in a mouse model. Nick has three mouse models.
Two are gene trap models, which means they can turn the GLDC gene on and off (with magic science). One model completely prevents GLDC gene expression, and the other allows approximately 10% of the GLDC gene expression, which allows the range of symptoms we see in NKH. The third is a missense model, which has the exact same mutation as a child living with NKH. This isn’t a gene they can turn on and off, these mice have NKH in the same way as our kids do.
Prof. Nick has two treatment projects at this ‘proof of concept’ stage. It’s very very exciting. They’re both in gene therapy – what this means is he’d like to place a working gene into a cell that is currently has a broken gene (thanks to an NKH mutation). What that means is the cell will be able to produce a stable protein, which brings the whole glycine cleavage system up online. Hallelujah! Also, to clarify, this is an effective treatment for EVERY child with a mutation in that gene. Whether it’s a missense mutation or a deletion.
The tricky bit is getting the gene into the cell. They use fancy technology called vectors, which are actually viruses with the bad stuff taken out (it’s kind of like hijacking our immune system. Viruses attach to our cells, depositing the bad stuff that makes us sick. Our body fights them off, and writes a little reminder of how to avoid that particular virus in our genes). Instead of depositing bad stuff, the virus will deposit a working gene!
None of this is new, by the way. Nick’s team are working on it, and have been working on it for the last three years – which is so important for people to know. There’s a charity that’s fundraising millions to REPEAT this work that’s already been done. If you’re in the community you’ll know who, and I suspect the reason they’re doing it is so they can direct the clinical trial specifically to their kids, but for me, it feels like a waste of money, repeating existing research.
I don’t want to fundraise (which is really really hard work, btw) to have existing research replicated. I want to fundraise for the team that’s going to get to clinical trial the fastest, which bluntly, is Prof. Nick’s team. It’s why we support Joseph’s Goal, as the only charity that supports Prof. Nick.
Now, Prof. Nick’s team don’t have gene replacement therapy down yet. They’re exploring with two different vectors – AAV vectors to target the brain and lentiviral vectors to target the liver (AAV + Lentiveral are just different kinds of viruses. Like say the man flu + regular flu!). There are many questions – which is the best vector to use? Is one enough, just the brain or just the liver? Do they need to do both at the same time? Will it work? Will it be safe?
This is why they need to a do a proof of concept. They have the mouse model ready to go (a huge undertaking in itself) but it’s going to cost roughly ~£2.5 million to get through to clinical trials, and around 2-3 years, if they’re funded, and if the safety and ethical regulators who patrol this type of work are kind.
Speaking of, the second step in getting treatment to clinical trial is the safety step. The team needs to show that the treatment is able to be ‘produced’ at a scale appropriate for patients the world over (meaning, safe for humans. It’s what they call ‘clinical grade’). It requires plenty of safety studies to demonstrate this.
Prof. Nick’s team have another project to do small molecule work that is almost at this point. ‘Small molecule work’ sounds very high level science – but the idea is that there will be a pill (or powder?) of some sort that might be more effective, or might have less side effects (we hope!) than the existing treatments (aka, Sodium Benzoate). The study they’ve done in the mice has been very very positive, and everyone is cautiously optimistic.
Nick is currently talking to regulators about what kind safety studies they would need to do to get to this point. He’ll need a study to confirm it’s effective (costing ~£35K), and another study to check for long-term safety/possible side effects (costing another ~£30k) which might take 12-18 months. He’ll also need to find a clinical grade (read: safe!) version of the medication to test in the trials.
Then, the third step is to go to a clinical trial. May it happen sooner rather than later.
So, in summary Prof Nicks team are:
- Using mass spectrometry (and other tests) to follow glycine around the body and understand what happens when there is too much glycine
- Trialling two different vectors (one of the liver, one for the brain) in gene replacement therapy as a treatment option
- Trialling small molecule work as a possible alternative to sodium benzoate.
It seems crazy to me that a proper, effective treatment (gene replacement therapy) is less than £3 million. That all that’s standing between us and a significantly improved quality of life is MONEY. It’s also not even that far out of reach, is it? If every person in London donated a £1, we’d have the funds three times over.
So. We fundraise. You know we’re fundraising. We’re cutting our hair. We’re eating chicken nuggets. We’re doing an NKH Race Night. We might raise between £5k – £6k between those three fundraisers (which awkwardly all happen on the same two days).
That’s approximately 2% of whats needed, but I genuinely feel like at least I’m making the effort. I’m trying. With all the desperation of a special needs mama who wants a future with her son. I’m two feet all in, desperately trying to fundraise. This past week we managed to raise almost £8k with our fundraisers that happened across the weekend. A mere drop in the ocean. Hey ho, onwards we go.